What is Cellular Aging? The Complete Guide to DNA Damage and Genomic Instability
Have you ever wondered why you feel more tired as you get older, why recovery from exercise takes longer, or why fine lines seem to appear out of nowhere? These common experiences are often dismissed as just "getting older," but they are symptoms of a deeper, invisible process happening at the cellular level. Recent groundbreaking research has identified the precise mechanisms behind aging, revealing that it's not simply the passage of time, but rather the accumulation of specific types of cellular damage that drives the aging process.
Since 2013, when the first comprehensive framework for understanding aging was published, close to 300,000 scientific articles have been published on this subject—as many as during the entire preceding century. This explosion of research has revealed that aging is driven by twelve interconnected biological processes, called the "hallmarks of aging," with DNA damage and cellular senescence at the very center of this complex web.
Key Takeaways
- Massive Daily DNA Assault: Your DNA endures up to one million damage events per cell every day, with normal cells accumulating over 2,000 mutations by middle age.
- Zombie Cell Explosion: Senescent "zombie cells" accumulate 2-20 fold from young adulthood to old age, secreting inflammatory signals that damage healthy tissues.
- 12 Interconnected Hallmarks: Aging is driven by twelve interconnected biological processes, from genomic instability to chronic inflammation.
- Repair is Possible: Your body has sophisticated DNA repair mechanisms that can be supported and enhanced to slow the aging process.
The Science of Aging: DNA Damage, Zombie Cells & The 12 Hallmarks: A Quick Overview
Before we dive deep into the the science of aging, watch this quick overview explaining the causes of cellular aging.

In This Comprehensive Guide:
- What is Cellular Aging? The Biological Reality Behind Getting Older
- The 12 Hallmarks of Aging: A Complete Framework
- The DNA Damage Crisis: Your Cellular Blueprint Under Attack
- Genomic Instability: When Repair Systems Fail
- Cellular Senescence: The Zombie Cell Phenomenon
- Inflammaging: How Chronic Inflammation Accelerates Aging
- The Interconnected Nature of Aging Processes
What is Cellular Aging? The Biological Reality Behind Getting Older
Cellular aging is the gradual decline in the health and function of our cells over time, driven by the accumulation of molecular damage and the progressive failure of cellular maintenance systems. Unlike the simple passage of chronological time, biological aging is an active process characterized by specific, measurable changes at the molecular, cellular, and systemic levels.
Think of cellular aging like a photocopier making copies of copies—with each new copy, the quality degrades slightly due to accumulated errors and wear on the machine. Our cells operate similarly, but the process is far more complex and involves multiple interconnected systems that can either accelerate or slow the aging process depending on various factors.
The distinction between chronological age (how many years you've lived) and biological age (how much your cells have aged) is crucial. Some people have biological ages significantly younger or older than their chronological age, which explains why some 60-year-olds have the energy and health of 40-year-olds, while others seem much older than their years.
The 12 Hallmarks of Aging: A Complete Framework
Modern aging research has identified twelve fundamental biological processes that drive aging. These "hallmarks" are interconnected and reinforce each other, creating a complex web of cellular dysfunction that manifests as the aging we experience. Understanding these hallmarks is crucial because they represent potential intervention points where we might slow or reverse aspects of the aging process.
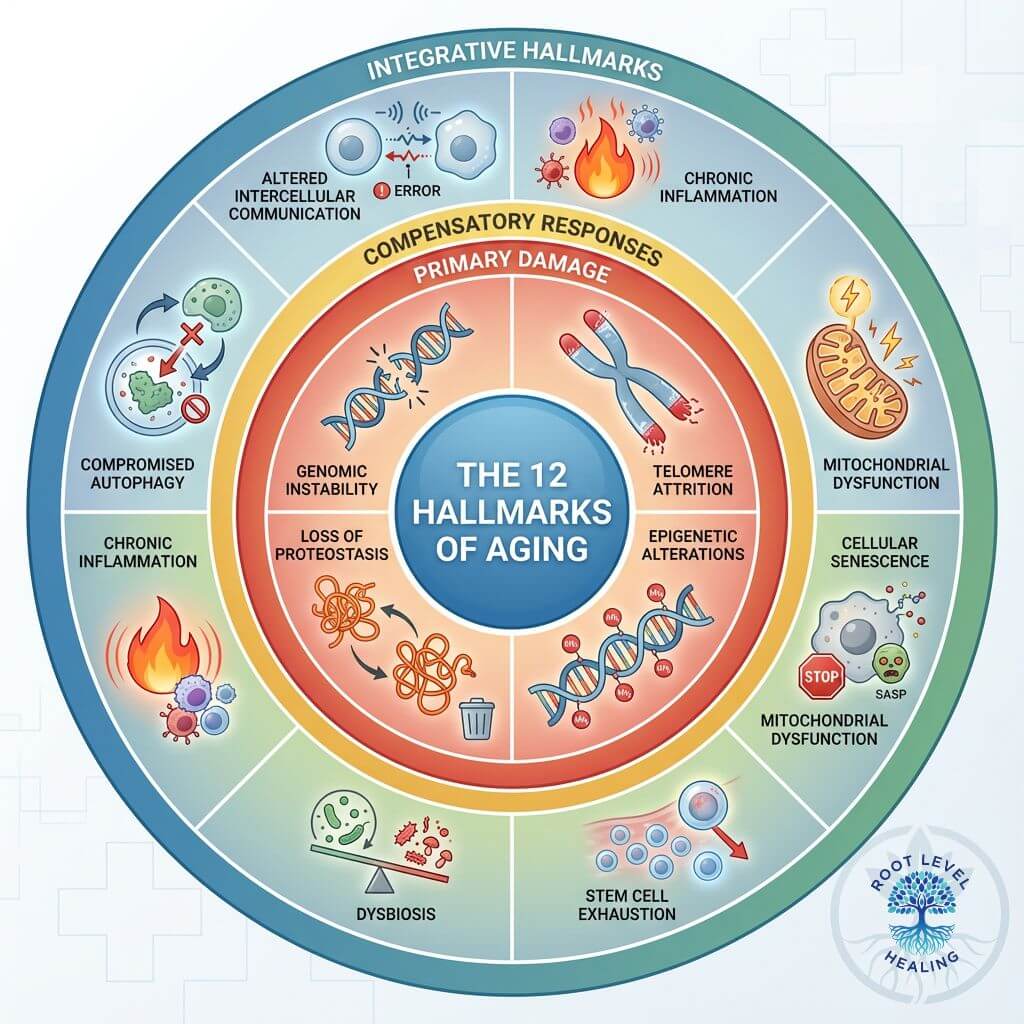
The twelve hallmarks of aging are interconnected processes that collectively drive the aging of our cells and tissues.
| Hallmark Category | Primary Hallmarks | Key Impact |
|---|---|---|
| Primary Damage | Genomic Instability, Telomere Attrition, Epigenetic Alterations, Loss of Proteostasis | Root causes of cellular damage |
| Compensatory Responses | Disabled Macroautophagy, Deregulated Nutrient-Sensing, Mitochondrial Dysfunction, Cellular Senescence | Body's attempts to manage damage |
| Integrative Hallmarks | Stem Cell Exhaustion, Altered Intercellular Communication, Chronic Inflammation, Dysbiosis | System-wide aging manifestations |
These hallmarks don't operate in isolation. For example, genomic instability can trigger cellular senescence, which then contributes to chronic inflammation, which in turn accelerates further genomic damage—creating a vicious cycle that accelerates aging throughout the body.
The DNA Damage Crisis: Your Cellular Blueprint Under Attack
At the heart of every cell lies your DNA—the master blueprint containing the instructions for everything your body does. This intricate genetic code faces an unprecedented daily assault that most people never realize is happening. The scale of this attack is staggering: scientists estimate that the DNA in a single human cell sustains up to one million damage events per day [1].
To put this in perspective, if your DNA were a library containing all the instructions for your body, it would be as if someone were randomly tearing pages, scribbling over text, and rearranging chapters a million times every single day in every library in a city of 37 trillion libraries (the approximate number of cells in your body).
The Sources of Daily DNA Damage
DNA damage comes from both external and internal sources, creating a constant barrage that your cellular repair systems must address:
External Threats:
- Ultraviolet radiation from sunlight, which can cause DNA cross-linking and mutations
- Environmental toxins including heavy metals, pesticides, and industrial chemicals
- Air pollution containing particulate matter and reactive compounds
- Lifestyle factors such as smoking, excessive alcohol consumption, and poor diet
- Ionizing radiation from medical procedures, cosmic rays, and natural background radiation
Internal Threats:
- Reactive oxygen species (ROS) generated during normal cellular metabolism
- DNA replication errors that occur during cell division
- Spontaneous chemical reactions such as DNA base deamination and depurination
- Metabolic byproducts that can react with and damage DNA
- Inflammatory processes that generate DNA-damaging compounds
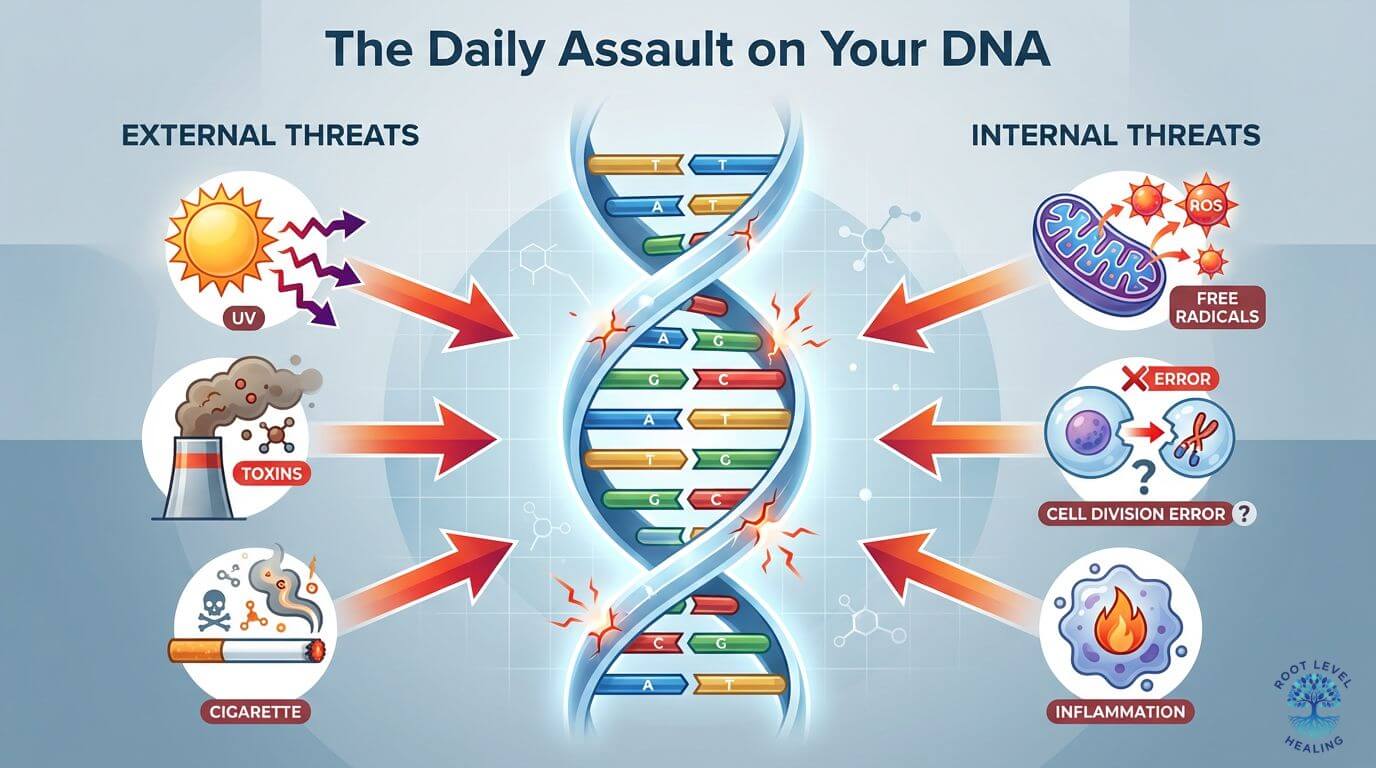
Our DNA is under constant assault from both external environmental factors and internal metabolic processes.
The Accumulation of Mutations Over Time
The cumulative effect of this daily damage is remarkable. Research has shown that normal esophageal epithelium cells from young individuals already display hundreds of mutations, and by middle age, these same cell types may carry more than 2,000 mutations per cell [2]. This represents a dramatic accumulation of genetic changes that occurs even in healthy, non-cancerous tissue.
Interestingly, this mutational burden varies significantly between different tissues and cell types. Rapidly dividing cells, such as those in the skin and digestive tract, tend to accumulate mutations faster than slowly dividing cells like neurons. However, even post-mitotic cells that rarely divide still accumulate DNA damage over time, contributing to age-related dysfunction in organs like the brain and heart.
Genomic Instability: When Repair Systems Fail
Fortunately, evolution has equipped our cells with sophisticated "molecular repair crews"—specialized enzymes and protein complexes that constantly patrol our DNA, identifying and fixing damage. These repair systems are remarkably efficient, handling the vast majority of daily DNA damage before it can cause problems. However, as we age, these repair mechanisms face increasing challenges.
The DNA Repair Arsenal
Your cells employ multiple DNA repair pathways, each specialized for different types of damage:
- Base Excision Repair (BER) - Fixes small, non-helix-distorting base lesions
- Nucleotide Excision Repair (NER) - Removes bulky DNA adducts and cross-links
- Mismatch Repair (MMR) - Corrects replication errors and small loops
- Homologous Recombination (HR) - Repairs double-strand breaks using sister chromatids
- Non-Homologous End Joining (NHEJ) - Directly ligates broken DNA ends
Age-Related Decline in Repair Efficiency
As we age, these repair systems become less efficient due to several factors:
Decreased Repair Protein Expression: Many key DNA repair proteins show reduced expression levels with age, limiting the cell's repair capacity.
Accumulated Repair System Damage: The repair machinery itself can become damaged over time, creating a vicious cycle where damaged repair systems are less able to fix new damage.
Energy Depletion: DNA repair is an energy-intensive process requiring significant amounts of ATP. Age-related mitochondrial dysfunction can limit the energy available for repair processes.
Chromatin Changes: Age-related alterations in chromatin structure can make DNA less accessible to repair machinery, slowing repair processes.
The Role of Sirtuins in DNA Repair
One particularly important family of proteins in DNA repair and aging is the sirtuins, especially SIRT6. Research has shown that SIRT6 plays a crucial role in maintaining genomic stability by:
- Facilitating double-strand break repair
- Regulating chromatin structure to allow repair access
- Coordinating the cellular response to DNA damage
- Modulating inflammation in response to genomic instability
Remarkably, overexpression of SIRT6 in mice has been shown to reduce genomic instability, improve DNA repair efficiency, and extend lifespan [3]. This suggests that enhancing the function of key repair proteins could be a viable strategy for slowing aging.
Cellular Senescence: The Zombie Cell Phenomenon
When DNA damage becomes too severe for a cell to repair, it faces a critical decision point. The cell can either self-destruct through a process called apoptosis (programmed cell death) or enter a state of permanent growth arrest called cellular senescence. Senescent cells, often referred to as "zombie cells," represent one of the most important discoveries in aging research.
The Senescent Cell Dilemma
Senescent cells are metabolically active but have permanently stopped dividing. While this prevents them from becoming cancerous (which would require continued cell division), it creates a different problem: these cells refuse to die and instead linger in tissues, where they cause significant harm to their neighbors.
The accumulation of senescent cells with age is dramatic. Research shows that senescent cells increase 2- to 20-fold when comparing young adults (under 35) to older adults (over 65) [4]. This accumulation affects multiple cell types, including fibroblasts, endothelial cells, and immune cells, and occurs in virtually all tissues throughout the body.
The Senescence-Associated Secretory Phenotype (SASP)
The real problem with senescent cells isn't just that they stop functioning properly—it's what they actively secrete. Senescent cells develop what researchers call the Senescence-Associated Secretory Phenotype (SASP), releasing a cocktail of inflammatory molecules, growth factors, and tissue-remodeling enzymes.
This SASP includes:
- Pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α
- Chemokines that attract immune cells and promote inflammation
- Matrix metalloproteinases that break down surrounding tissue structure
- Growth factors that can promote abnormal cell growth
- Damage-associated molecular patterns (DAMPs) that trigger immune responses
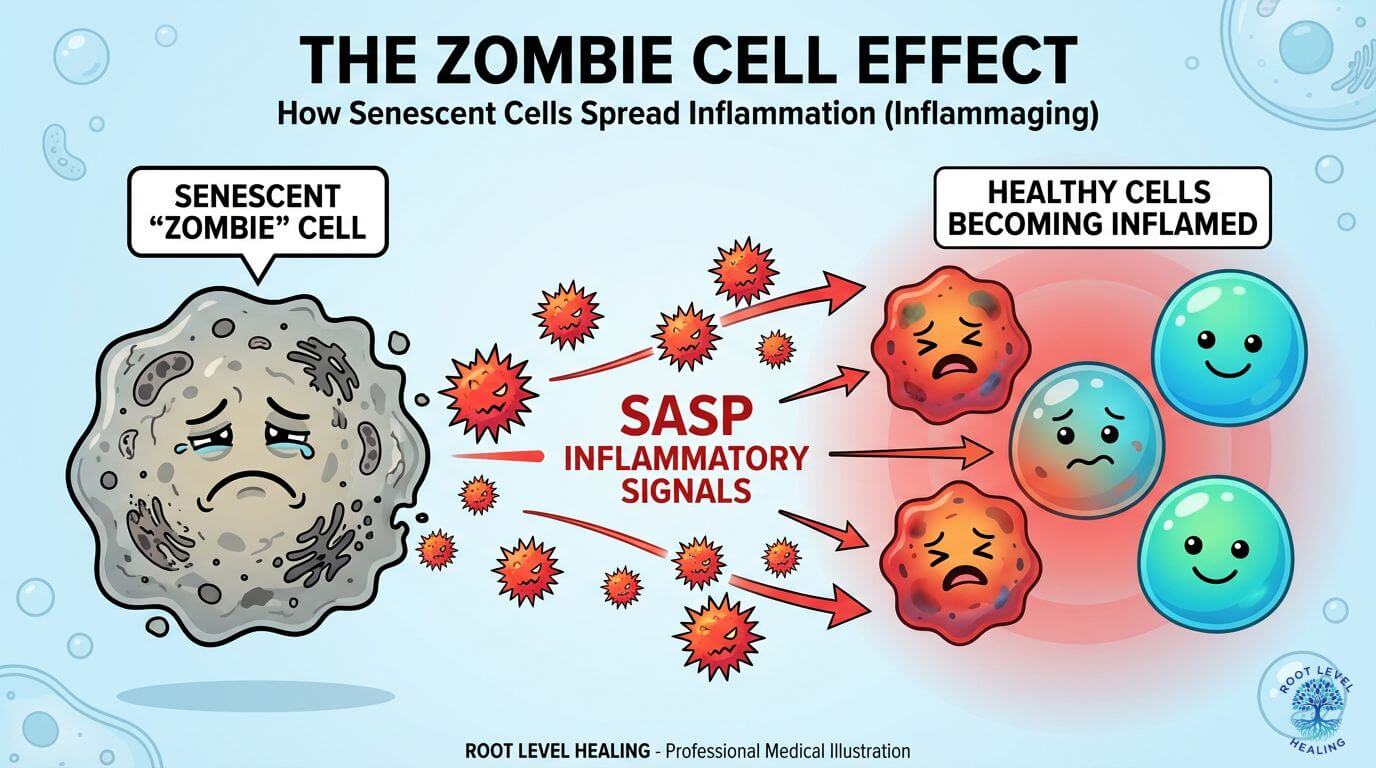
Senescent "zombie" cells create a toxic, inflammatory environment that damages healthy neighboring cells, accelerating the aging process.
The Spreading Effect of Senescence
Perhaps most concerning is that senescence can be "contagious." The inflammatory signals released by senescent cells can induce senescence in nearby healthy cells, creating a spreading wave of cellular dysfunction. This paracrine senescence helps explain why aging tends to accelerate over time—as more cells become senescent, they create an increasingly toxic environment that promotes senescence in their neighbors.
Inflammaging: How Chronic Inflammation Accelerates Aging
The accumulation of senescent cells and their inflammatory secretions contributes to a phenomenon that researchers have termed "inflammaging"—a chronic, low-grade inflammatory state that characterizes aging. This isn't the acute inflammation you experience with an injury or infection, which is beneficial and resolves quickly. Instead, inflammaging is a persistent, systemic inflammation that damages tissues and accelerates aging processes throughout the body.
The Vicious Cycle of Inflammation and Aging
Inflammaging creates a self-perpetuating cycle:
- Initial Damage: DNA damage and cellular stress trigger inflammatory responses
- Senescent Cell Accumulation: Damaged cells become senescent and secrete inflammatory factors
- Systemic Inflammation: SASP factors enter circulation, promoting inflammation throughout the body
- Further Damage: Chronic inflammation causes additional DNA damage and cellular stress
- Accelerated Aging: The cycle repeats and intensifies, accelerating aging processes
Health Consequences of Inflammaging
Chronic inflammation has been linked to virtually every age-related disease, including:
- Cardiovascular disease through endothelial dysfunction and atherosclerosis
- Neurodegeneration including Alzheimer's and Parkinson's disease
- Metabolic dysfunction such as type 2 diabetes and metabolic syndrome
- Cancer through DNA damage and tissue remodeling
- Osteoporosis via inflammatory effects on bone metabolism
- Immune system decline leading to increased infection risk
The Interconnected Nature of Aging Processes
One of the most important insights from modern aging research is that the twelve hallmarks of aging are not independent processes—they are deeply interconnected and mutually reinforcing. This interconnectedness explains why aging tends to accelerate over time and why interventions targeting multiple hallmarks simultaneously may be more effective than those targeting individual processes.
Examples of Hallmark Interconnections
Genomic Instability → Multiple Hallmarks:
- DNA damage can trigger cellular senescence as a protective mechanism
- Mutations in epigenetic regulators lead to epigenetic alterations
- Damaged genes encoding proteins cause loss of proteostasis
- DNA leakage into the cytoplasm triggers chronic inflammation
- Mutations affecting nutrient-sensing pathways cause deregulated nutrient-sensing
Cellular Senescence → Systemic Effects:
- SASP factors promote chronic inflammation throughout the body
- Inflammatory signals disrupt intercellular communication
- Senescent stem cells lead to stem cell exhaustion
- Tissue dysfunction affects microbiome balance (dysbiosis)
Therapeutic Implications
Understanding these interconnections has important implications for anti-aging interventions. Rather than targeting individual hallmarks in isolation, the most effective approaches are likely to be those that address multiple interconnected processes simultaneously. This systems-level approach to aging intervention represents the future of longevity medicine.
The interconnected nature of aging also explains why lifestyle interventions that seem simple—such as regular exercise, caloric restriction, and stress management—can have such profound effects on aging. These interventions simultaneously impact multiple hallmarks, creating beneficial cascades throughout the aging network.
As we delve deeper into the mechanisms of aging, it becomes clear that the process is both more complex and more hopeful than previously thought. While aging involves multiple interconnected processes, this same interconnectedness means that interventions targeting key nodes in the aging network could have widespread beneficial effects. In our next article, we'll explore one of the most promising intervention targets: NAD+, the "fountain of youth molecule" that powers many of the cellular repair processes we've discussed.
Frequently Asked Questions
Q: What exactly is cellular aging and how does it differ from chronological aging?
A: Cellular aging refers to the biological deterioration of cells over time, characterized by accumulated DNA damage, protein dysfunction, and cellular senescence. Unlike chronological aging (simply the passage of time), biological aging can vary significantly between individuals and can be influenced by lifestyle, genetics, and environmental factors.
Q: How much DNA damage occurs daily in our cells?
A: Each cell in your body experiences up to one million DNA damage events per day. By middle age, normal cells can accumulate over 2,000 mutations. This damage comes from both external sources (UV radiation, toxins) and internal processes (metabolism, replication errors).
Q: What are the 12 hallmarks of aging?
A: The 12 hallmarks are: genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, disabled macroautophagy, deregulated nutrient-sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, altered intercellular communication, chronic inflammation, and dysbiosis.
Q: What are "zombie cells" and why are they harmful?
A: Zombie cells (senescent cells) are damaged cells that stop dividing but refuse to die. They secrete inflammatory signals called SASP that damage surrounding healthy cells. These cells accumulate 2-20 fold from young adulthood to old age, contributing to chronic inflammation and accelerated aging.
Q: Can cellular aging be slowed down or reversed?
A: Yes, research shows that supporting DNA repair mechanisms, reducing oxidative stress, eliminating senescent cells, and maintaining healthy lifestyle habits can significantly slow cellular aging. Some interventions have even shown reversal of certain aging markers in laboratory studies.
Q: What causes genomic instability?
A: Genomic instability occurs when DNA repair mechanisms become overwhelmed by damage from free radicals, environmental toxins, UV radiation, metabolic processes, and replication errors. Age-related decline in repair efficiency exacerbates this instability.
Q: How are the hallmarks of aging interconnected?
A: All 12 hallmarks are deeply interconnected. For example, genomic instability can trigger cellular senescence, which promotes chronic inflammation, which causes further DNA damage. This creates reinforcing cycles that accelerate aging throughout the body.
Q: What is inflammaging and how does it affect health?
A: Inflammaging is chronic, low-grade inflammation that characterizes aging. It's caused by senescent cells and other aging processes, and contributes to virtually every age-related disease including cardiovascular disease, neurodegeneration, diabetes, and cancer.
Q: At what age does cellular aging accelerate?
A: While cellular aging begins early in life, it typically accelerates around age 40-50 when DNA repair efficiency starts declining more rapidly, senescent cells begin accumulating significantly, and multiple aging hallmarks start reinforcing each other.
Q: What role do sirtuins play in aging?
A: Sirtuins, particularly SIRT6, are crucial for DNA repair and genomic stability. They help coordinate cellular responses to damage and regulate inflammation. Research shows that enhancing sirtuin function can reduce genomic instability and extend lifespan in laboratory animals.
References
- De Bont, R., & van Larebeke, N. (2004). Endogenous DNA damage in humans: a review of quantitative data. Mutagenesis, 19(3), 169-185.
- López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M., & Kroemer, G. (2023). Hallmarks of aging: An expanding universe. Cell, 186(2), 243-278.
- Tian, X., Firsanov, D., Zhang, Z., et al. (2019). SIRT6 is responsible for more efficient DNA double-strand break repair in long-lived species. Cell, 177(3), 622-638.
- Olivieri, F., Rippo, M. R., Monsurrò, V., et al. (2013). MicroRNAs linking inflamm-aging, cellular senescence and cancer. Ageing Research Reviews, 12(4), 1056-1068.
- Schumacher, B., Pothof, J., Vijg, J., & Hoeijmakers, J. H. (2021). The central role of DNA damage in the ageing process. Nature, 592(7856), 695-703.
Ready to Discover Your Score?
This 2-minute quiz is the essential first step to understanding your body's current toxic load and identifying the root cause of how you feel.
Take The Toxicity QuizExplore More Articles
Dive deeper into topics like detoxification, gut health, and mental clarity on our blog. Knowledge is the key to taking control of your health.
Visit The Blog